Our Translational Research Framework
From in vitro screening to clinical trials, our platform unites data, modeling, and collaboration to accelerate the development of effective TB drug regimens.




.avif)
.avif)
The TB Regimen Challenge
Developing TB treatments is especially complex. With tens of thousands of possible drug combinations, traditional trial-and-error methods are too slow and costly.
Discover the Innovations at Our Labs

Pediatric Focus Committee (PFC)
Children are typically not considered until adult drug development is complete, leading to pediatric dosing recommendations that rely on limited data and often overlook principles of developmental pharmacology. Our work seeks to strengthen both early- and late-phase pediatric TB drug development by: (a) predicting pediatric dosing for new molecules early in the development pathway; (b) revising recommendations for existing drugs that yield unacceptably poor treatment outcomes in certain subgroups of children; (c) identifying high-burden populations in need of treatment optimization, including children who are HIV-positive, malnourished, or very young; (d) using real-world data to predict pharmacokinetics in representative pediatric populations; and (e) applying model-based approaches and prior trial data to design smarter, more efficient pediatric trials. Ensuring that children with TB benefit from advances in TB therapeutics as early as possible is critically important. The Pediatric Focus Committee (PFC) brings together experts in developmental immunology, pediatric TB biomarkers, developmental pharmacology and PK modeling, first-time-in-pediatric and PK-safety studies, and pediatric formulations to achieve these goals.
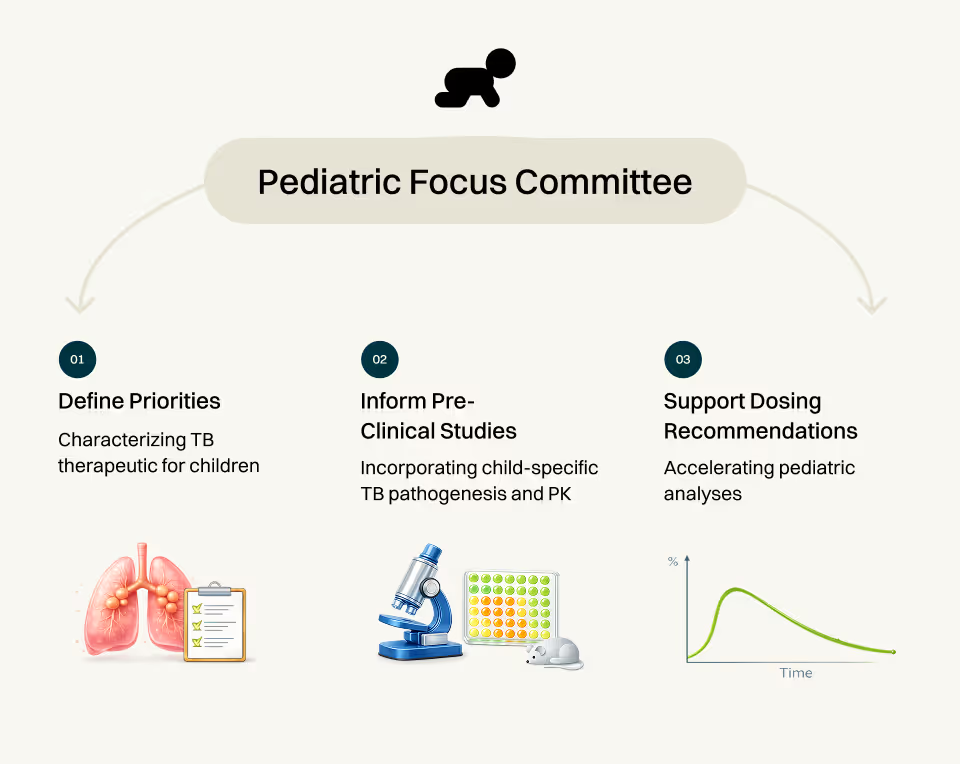
Pediatric Focus Committee (PFC)
Who we are: We are experts in pediatric tuberculosis, and lead research in immunology, biomarkers, diagnostics, pharmacology, and clinical trials to improve the care of children with TB.
Our goals: Children are typically not considered until adult drug development is complete, leading to pediatric dosing recommendations that rely on limited data and often overlooked principles of developmental pharmacology. We thus seek to accelerate the development of new TB therapeutics and regimens that consider child-specific needs.
Our approach: We serve as a cross-functional partner with the pre-clinical lab groups and data science and modeling group. We do this by 1) Defining TB therapeutic requirements and priorities for children; 2) Providing recommendations on how to incorporate the unique pathogenesis and pharmacokinetics of childhood TB into pre-clinical studies; and 3) Supporting pediatric analyses to guide dosing recommendations for children.
What Powers Our Approach
 High impact Preclinical Research
High impact Preclinical Research Data Science and Computational Modeling
Data Science and Computational Modeling Predictive Tools
Predictive Tools
Scientific Research Core
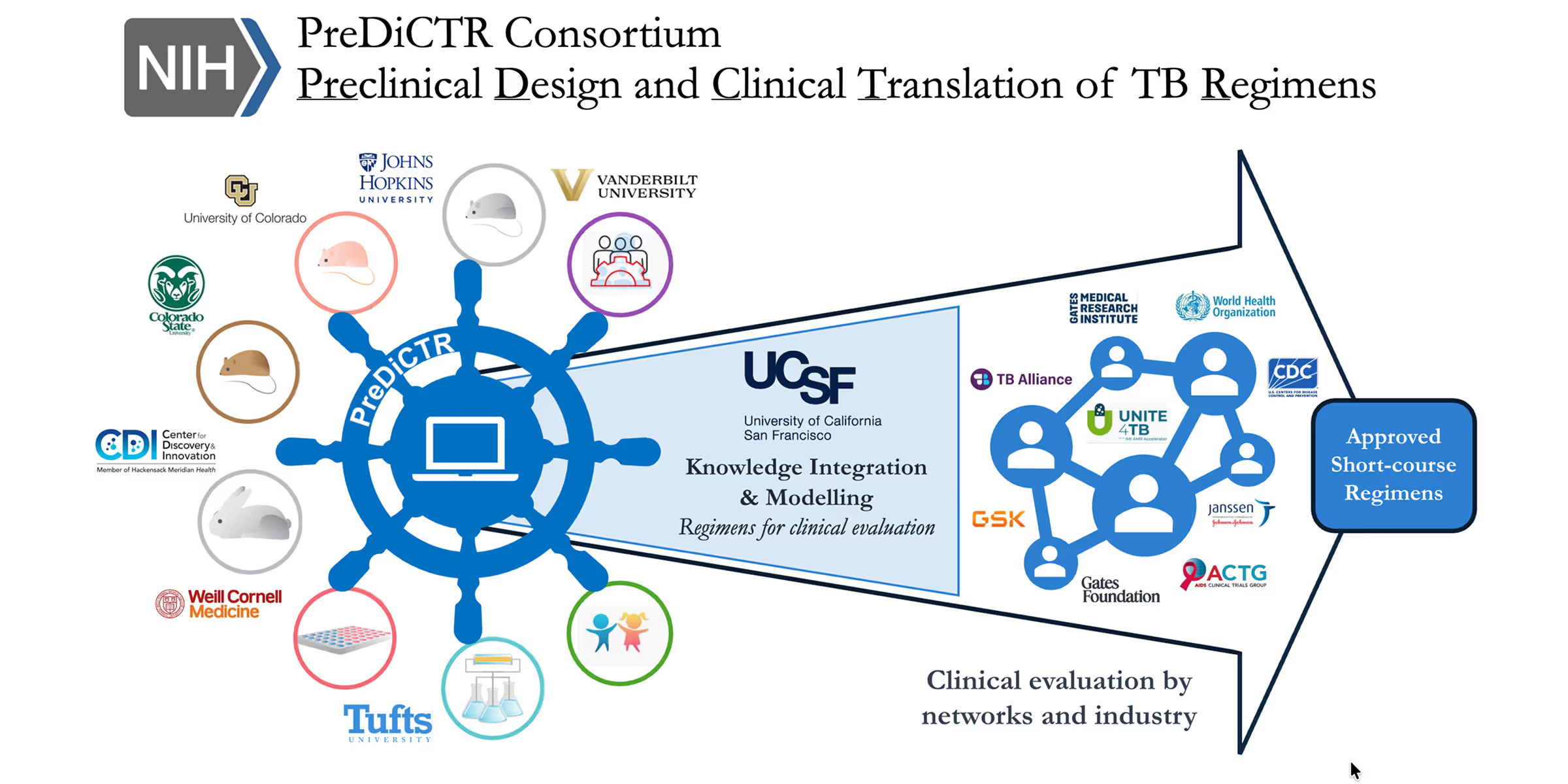
The PReDiCTR-TB Consortium integrates advanced technologies developed by the core group’s preclinical labs, including: highly predictive TB mouse models and an ultra-short-course murine model to assess treatment-shortening in one month (JHU/CSU Mouse PL); LCM-based drug distribution analysis and ex vivo caseum potency models (HMH CDI DMPK PL); physiologic, culture-independent potency biomarkers such as the RS ratio (CU PhyBM PL); large-scale empirical assays of combination drug responses in Mtb (TU DiaMOND PL); metabolomic and chemogenomic platforms to study drug action and resistance (WCM/RU Omics PL); and advanced data science and modeling tools (UCSF DSMG).




See How Our Consortium Aims to Support Clinical Trials








.avif)
.avif)
.avif)

.avif)
.avif)
.avif)
.svg)










%20(1).avif)
.avif)
.avif)
%20(1).avif)
%20(1).avif)
%20(1).avif)
.avif)
%20(1).avif)
%20(1).avif)
%20(1).avif)
.avif)
