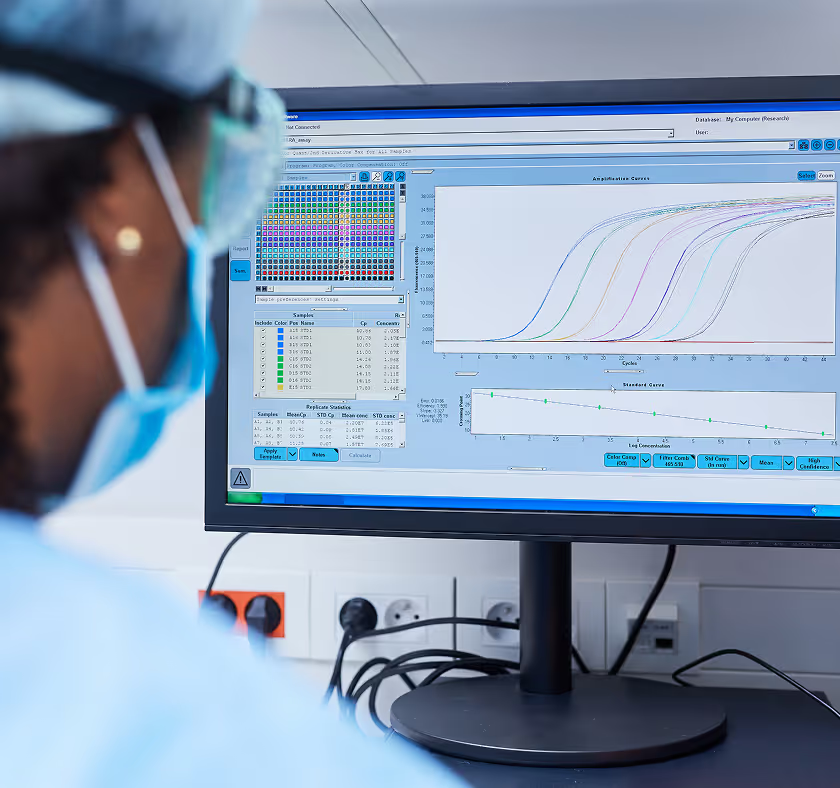
Revolutionizing tuberculosis regimen development
Abstract
Today's first-line tuberculosis regimen was developed in the 1950s to 1970s, followed by a long period of stagnation. New drugs have progressed to market only recently, with long timelines from target discovery to clinical trial success, alongside costly Phase 3 failures. Currently, the tuberculosis drug development pipeline is robust, containing multiple new chemical entities from diverse drug classes, motivating us to optimize this opportunity to advance compounds effectively and efficiently. In this article, we explore how recent innovations in data integration and computational methods are revolutionizing tuberculosis drug development, accelerating development timelines, and heightening the probability of success. We anticipate that these breakthroughs will lead to approval of novel drugs in unprecedented time frames, marking a significant milestone in the fight against this age-old disease. This progress is timely, as resistance to even recently registered drugs is emerging rapidly. Our hope is that these strategies will also be of value in other medical fields.

Related Publications

Emergence of antibiotic-specific Mycobacterium tuberculosis phenotypes during prolonged treatment of mice
Translational Modeling of BTZ-043 to Predict Phase 2A Efficacy and Evaluate Drug-Drug Interactions With Bedaquiline, Pretomanid, and Linezolid in Murine Models